
Preschool STEAM (for children ages 2-6) is all about having fun with science, math and art! There is no right or wrong way for children to experiment – the fun and learning comes from the doing! Early childhood research shows us that little hands and minds learn best when at play.
For our July STEAM adventure, we focused on creating an eruption ~ we called it Wizard’s Brew. We sought to create a chemical reaction by mixing vinegar and baking soda. For good measure, we also included some detergent and liquid watercolors.
The Question
Does combining vinegar and baking soda create a chemical reaction?
What happens when you add baking soda to vinegar? Does anything change when you add dishwashing detergent and liquid watercolors to the mix?
INGREDIENTS FOR OUR EXPERIMENT
Baking Soda
White vinegar
Liquid Watercolors
Plastic droppers
Plastic spoons
Plastic bowls
Mason jars
The Process
- Prior to the start of the program we made sure each table had two boxes of baking soda, 2 plastic spoons, 2 bottles of dishwashing detergent and 2 bottles of liquid watercolors. We did this to save on costs and encourage collaboration and sharing among the children.
- As each family arrived, we gave them a mason jar filled with 1/2 cup vinegar; we also gave them a small plastic bowl and a tray in which to place their jar.
- The initial instructions were to pour a small amount of liquid watercolor into the plastic bowl along with a dash of dishwashing liquid. That mixture was then set aside.
- Next the families were instructed to place a teaspoon (or more) of baking soda into the mason jar filled with vinegar.
- The result? An eruption!

- The result? An eruption!
- We then asked everyone to pour their mixture of liquid watercolor and dishwashing detergent into their mason jars and voila! There was a big, bubbly eruption in living color! What happened after that eruption? The fizz fizzled OUT!


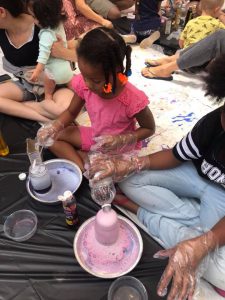
The Science Behind It
Carbonic Acid and Carbon Dioxide
“When baking soda is mixed with vinegar, a reaction takes place. The two chemicals, acetic acid and sodium bicarbonate, do not mix with each other; therefore causing a reaction. When the two chemicals are mixed, a new chemical is formed called carbonic acid. This chemical is very unstable and therefore it instantly starts falling apart and it turns into carbon dioxide and water. When the acetic acid and sodium bicarbonate meet, the carbonic acid is produced. It then falls apart thus leaving only carbon dioxide and water remaining. Bubbles begin floating around the top of the experiment. The bubbles, produced by this reaction, are actually the carbon dioxide escaping. Carbon dioxide is a gas and therefore does not mix with water. The carbon dioxide escapes from the water through the bubbles. Carbon dioxide is the air we exhale when we breathe.”
Sodium Acetate
“After all of the carbon dioxide escapes; the mixture suddenly stops bubbling, popping, and crackling. This happens because the carbon dioxide is what caused the reaction and once the carbon dioxide is gone, the reaction stops. After it stops, all that is left in the glass is sodium acetate and water.”
Credit: Village Bakery





















Join us on Monday, August 27 for our next Preschool STEAM program! Central Library ~ Studio on 4th, 10:30-11:30 a.m.