
Preschool STEAM (for children ages 2-6) is all about having fun with science, math and art! There is no right or wrong way for children to experiment – the fun and learning comes from the doing! Early childhood research shows us that little hands and minds learn best when at play. For our April STEAM adventure, we focused on oil and water. You know the saying: “Oil and water don’t mix!” Is there some truth to that saying? And if so, why???
The Question
Do oil and water mix? Yes or No
What happens when you add an emulsifier like dishwashing liquid to a mixture of oil and water?
INGREDIENTS FOR OUR EXPERIMENT
Credit: Childhood 101

Vegetable Oil
Water
Liquid watercolors
Plastic droppers
Plastic bowls
Dishwashing detergent
Mason Jars
The Process
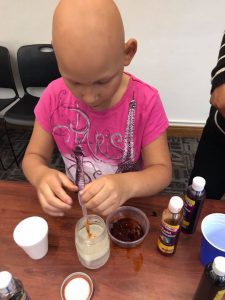
- We began by passing out 1/2 cup of oil, 1 cup of water and 1 mason jar to each family. The instructions were to pour 1 cup of water and 1/2 cup of oil into the mason jar, tightly seal the lid and just observe what happens. We also encouraged the children to shake their jars of liquid (no prompting necessary) to see what would happen.


- After quite a bit of shaking, everyone noticed that the oil and water kept separating with the water on the bottom and the oil right on top. We then moved to part 2 of our exploration and asked each family to get a small bowl of dishwashing detergent, a plastic dropper and a splash of some of the liquid watercolors we had on hand. The families then mixed the detergent and the liquid watercolor together. They then used their droppers to place the mixture into the glass jar with the oil and water.


- We allowed time for observation and play and encouraged everyone to notice what was happening to the mixture. Not only did the oil and water mix, each child had a beautiful work of art in a jar thanks to the addition of the liquid watercolors.

The Science
What happens when you mix oil and water?
Firstly oil and water do not mix. Even if you shake the jar the oil will immediately separate from the water as soon as it settles. Oil molecules are attracted to other oil molecules so they stick together. The same goes for water molecules….. so they just don’t mix – they are immiscible.
Secondly, the oil always floats on top of the water because the oil has a lower density than water.
Detergent is different again. It is attracted to both water and oil molecules. Detergent grabs onto both types of molecules causing oil droplets to be suspended in the water. When you shake the jar the detergent molecules adhere the water and oil together forming an emulsion. An emulsion is the combination of molecules that are not normally attracted to each other, that don’t usually mix. That is why detergent is so useful for cleaning greasy dishes!
Credit: Childhood 101
Budding Scientists at Work














We are taking the month of May off to come up with some cool experiments for the rest of the year! Join us for our next Preschool STEAM program on Monday, June 25 from 10:30-11:30 a.m. in Central Library’s Studio on 4th. No registration required! For ages 2-6!